
Top Benefits of Telehealth Implementation in Cognitive Healthcare
Following the COVID-19 pandemic, telehealth implementation...
Read more
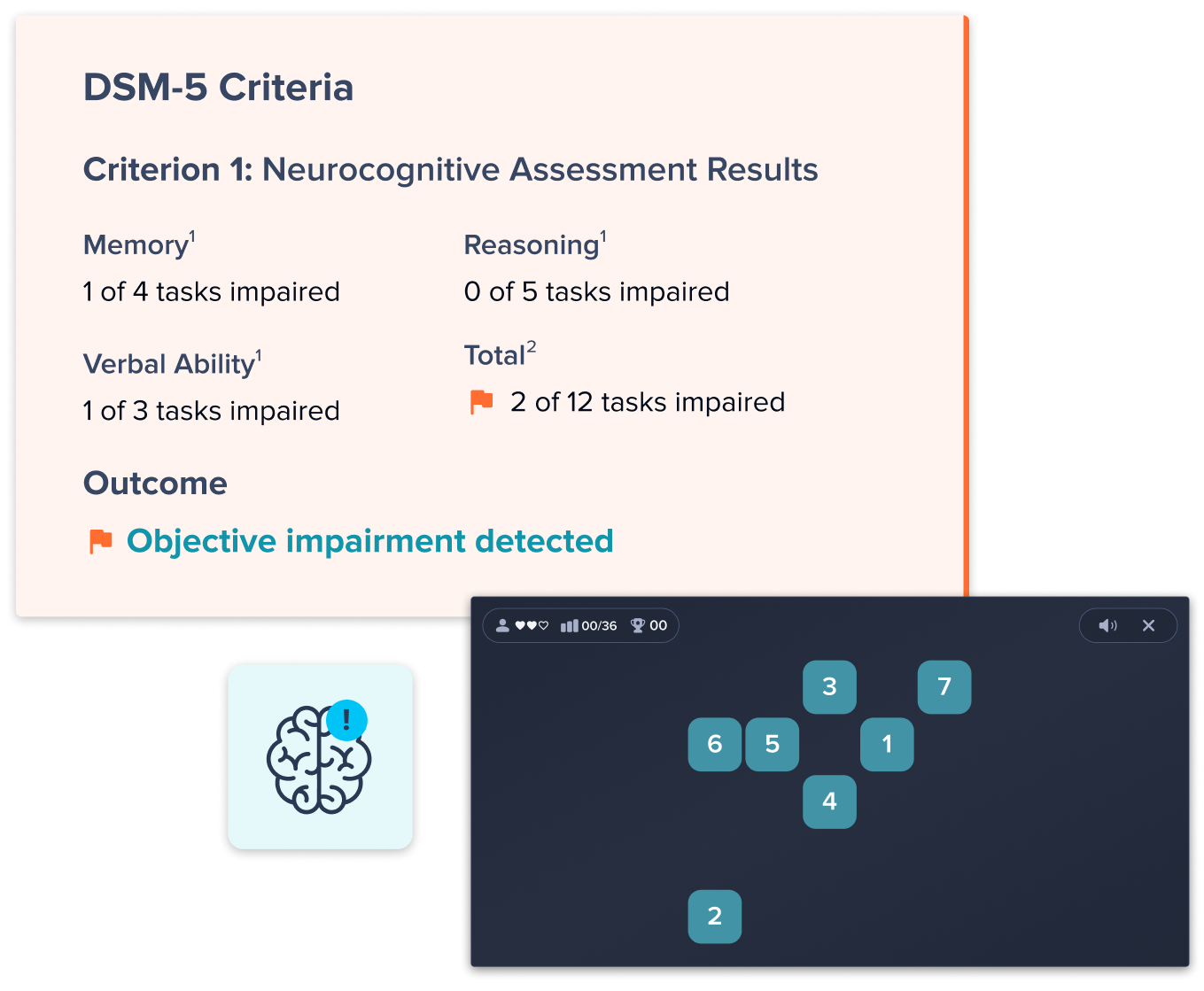
Decline is identified too late—after functional loss and avoidable high-cost events.
Unrecognized impairment leads to preventable ED visits, stays, and readmissions.
Cognitive barriers undermine adherence, self-management, and overall disease stability.
Early warning signs are overlooked, limiting timely, cost-saving action.

Undiagnosed dementia drives more emergency visits and hospital readmissions—many of which earlier identification can avoid.

~30% lower Alzheimer’s-related medical costs when mild cognitive impairment (MCI) is screened for early versus no screening.

Community dementia screening generates nearly 10% total cost savings over 10 years by preventing escalated care needs.

Earlier diagnosis delays progression to advanced disease stages, reducing long-term care utilization, complexity, and spend.
Plans and providers don’t need another standard screener. They need precise, actionable cognitive data to identify risk early and scale care across populations. Creyos enables providers and plans to:






Provide employed and affiliated physician groups with Creyos assessments to standardize testing, improve documentation accuracy, and capture data at scale to identify risk across your member population.

Administered in-clinic or remotely through existing EHR workflows, assessments fit into established visits, clinical operations, and population health reporting.

Patients complete a validated, self-administered assessment on a tablet or device, either in-clinic or remotely, that aligns with existing visit workflows to minimize burden on staff.
Creyos generates standardized, age-benchmarked cognitive results that providers can review directly and use to inform care decisions, documentation, and next-step planning.

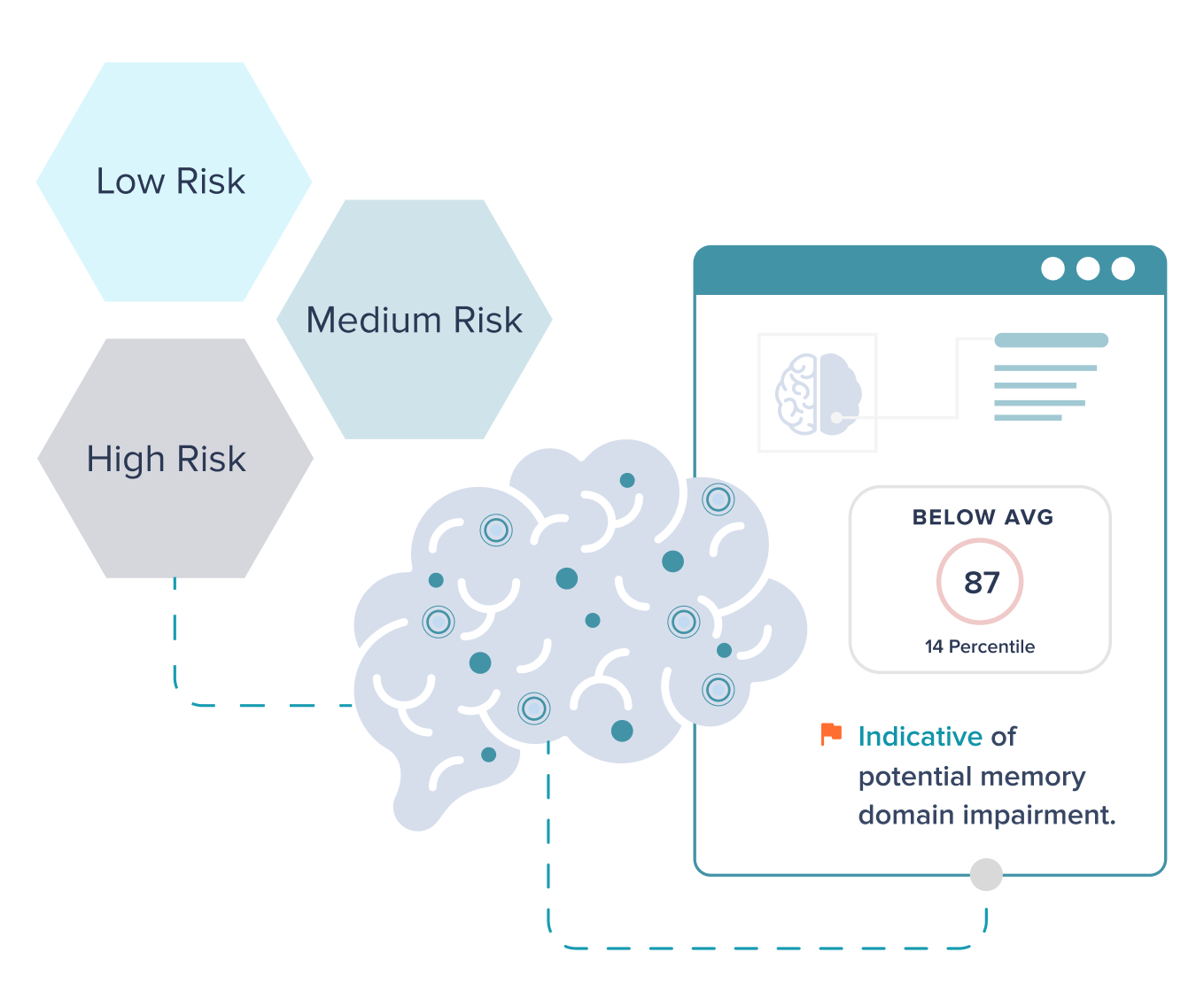
Nuanced and population-level cognitive data can feed plan-level initiatives such as risk stratification, resource prioritization, and longitudinal monitoring—without creating new reporting work.

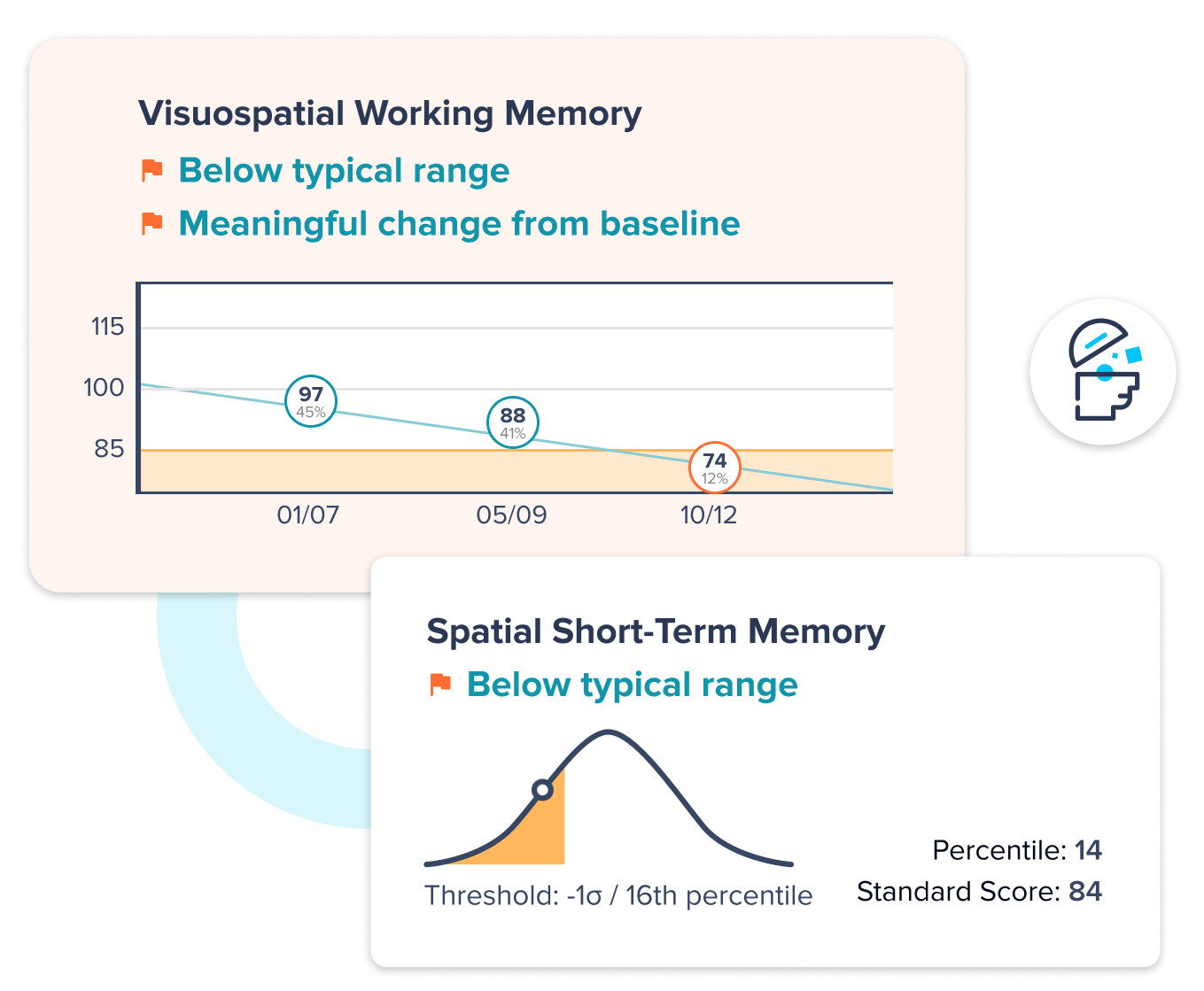
Providers can repeat assessments over time to observe cognitive change, helping plans and care teams proactively adjust support as patient needs evolve.
Creyos is based on decades of cognitive neuroscience research and validated across large, diverse populations. It provides domain-specific objective, interpretable cognitive data that supports more accurate clinical decision-making at the point of care.
1 Persons undiagnosed or unaware of dementia experience worse hospitalization outcomes. Source: Amjad, H., et al. (2025). Hospitalization outcomes among older adults living undiagnosed or unaware of dementia. Alzheimer’s & Dementia (Amsterdam, Netherlands), 17(1), e70051. https://doi.org/10.1002/dad2.70051
2 Increasing MCI screening coverage from 0% to about 80% significantly reduces total Alzheimer’s-related medical costs by approximately 29.83% versus no screening. Source: Zang, G. Y., Rao, K., Wu, A. T., Tang, Y., & Zhang, Z. (2025). Cognitive impairment screening strategy to reduce the burden of Alzheimer's disease in Shanghai: A system dynamics approach. Journal of Alzheimer's disease reports, 9, 25424823251337941. https://doi.org/10.1177/25424823251337941
3 Markov model simulations show community-based dementia screening can achieve ~9.83% total care cost savings over 10 years compared to no screening. Source: Saito, E., Nakamoto, B. K., Mendez, M. F., Mehta, B., & McMurtray, A. (2014). Cost effective community based dementia screening: a markov model simulation. International journal of Alzheimer's disease, 2014, 103138. https://doi.org/10.1155/2014/103138
4 Economic analyses emphasize the importance of identifying MCI early to improve patient and economic outcomes and reduce future care costs. Source: Frech, F. H., Li, G., Juday, T., Ding, Y., Mattke, S., Khachaturian, A., Rosenberg, A. S., Ndiba-Markey, C., Rava, A., Batrla, R., De Santi, S., & Hampel, H. (2024). Economic impact of progression from mild cognitive impairment to Alzheimer disease in the United States. The Journal of Prevention of Alzheimer’s Disease, 11(4), 983–991. https://doi.org/10.14283/jpad.2024.68

Following the COVID-19 pandemic, telehealth implementation...
Read more

Sleep, far from being a passive state of rest, is a dynamic...
Read more

According to a study from the Alzheimer’s Society, over 62%...
Read more